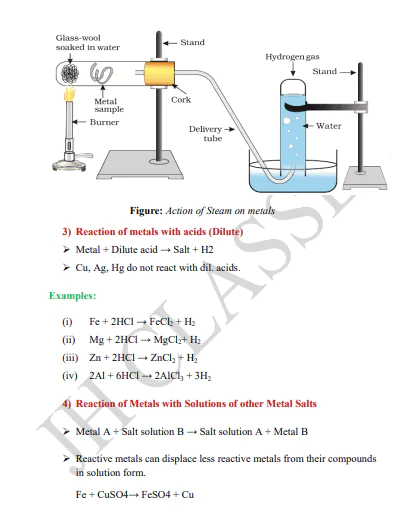
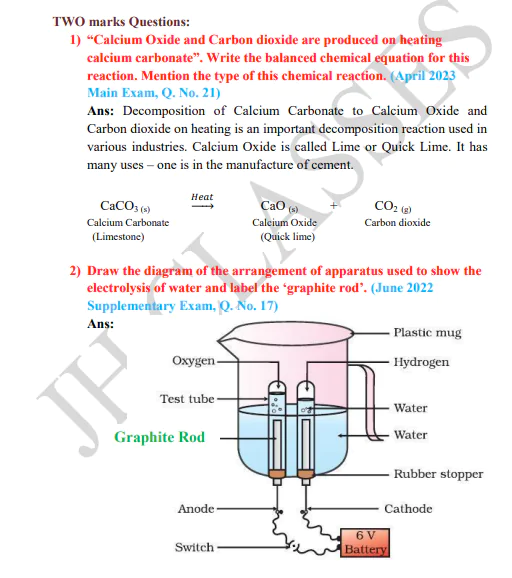
Description
Chemical reactions are processes where substances (reactants) undergo a transformation to form new substances (products) with different properties. These reactions involve the breaking and forming of chemical bonds. A chemical equation represents this change by showing the reactants and products using chemical formulas, with arrows indicating the direction of the reaction. The equation must be balanced to ensure that the number of atoms of each element is conserved on both sides of the equation, following the Law of Conservation of Mass.

















 Maharshi Dayanand University | B.E/B.Tech 3rd Semester Object Oriented Programming Notes PDF
Maharshi Dayanand University | B.E/B.Tech 3rd Semester Object Oriented Programming Notes PDF  Mathematics Notes for BA 3rd Semester Notes in English - Maharshi Dayanand University | Complete Printable Notes
Mathematics Notes for BA 3rd Semester Notes in English - Maharshi Dayanand University | Complete Printable Notes
Reviews
There are no reviews yet.