Description
Acids and bases are two types of chemical substances with distinct properties. Acids are substances that release hydrogen ions (H⁺) when dissolved in water, and they tend to have a sour taste and turn blue litmus paper red. Bases, on the other hand, release hydroxide ions (OH⁻) in water, and they are typically bitter, slippery, and turn red litmus paper blue. Acids and bases can neutralize each other, forming water and a salt in a process called neutralization. The strength of an acid or base is measured by its pH, with acids having a pH less than 7 and bases having a pH greater than 7.




















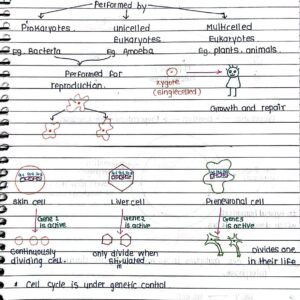
 Oedema PATHOLOGY BDS 2nd year handwritten notes for University exams PDF
Oedema PATHOLOGY BDS 2nd year handwritten notes for University exams PDF  Maharshi Dayanand University | MCA 2nd Semester Theory of Computation Notes in English - Complete Printable Notes
Maharshi Dayanand University | MCA 2nd Semester Theory of Computation Notes in English - Complete Printable Notes  Maharshi Dayanand University | MBA 4th Semester B2B Marketing Notes PDF
Maharshi Dayanand University | MBA 4th Semester B2B Marketing Notes PDF  HIV/AIDS MICROBIOLOGY BDS 2nd year handwritten notes for University exams
HIV/AIDS MICROBIOLOGY BDS 2nd year handwritten notes for University exams
Reviews
There are no reviews yet.